A Better Treatment Option for Patients with Chronic Constipation
ANJ908 (Pradigastat) has the potential to markedly improve treatment of chronic constipation. Patients currently cycle through several prescription medications without resolution of their disease.
Bridging the Treatment Gap
ANJ908 introduces a new mechanism to treat constipation that can potentially out-perform standard of care.
Discovery: A Better Target for Chronic Constipation
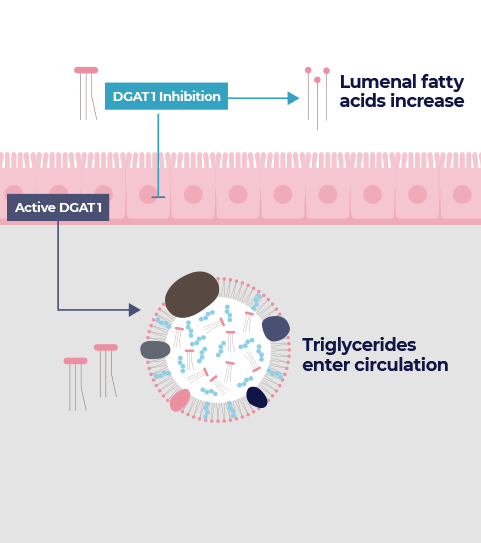
An enzyme called diacylglycerol acyltransferase 1 (DGAT1) was identified in the early 2000s as a possible target for metabolic diseases. DGAT1 plays a key role in the re-assembly of meal triglycerides, acting as a sensor of dietary fat intake.
In clinical trials, an unexpected observation was increased bowel movement frequency and softer stool. Anji is capitalizing on this consistent human pharmacology to realize the benefit of DGAT1 inhibition and better manage chronic constipation.
The inhibition of DGAT1 by ANJ908 changes the intestinal milieu, namely by raising the level of fatty acids in the gut lumen. This leads to increased colon motility and water secretion, two necessary attributes for effective constipation therapy.
ANJ908 Trials
To test the DGAT1 hypothesis, Anji has conducted a Phase 2 clinical PoC trial of ANJ908 (pradigastat) in patients with chronic idiopathic constipation. Patients in the U.S. and China were recruited to participate in a placebo-controlled, double-blind study to demonstrate the efficacy, safety, and tolerability of ANJ908.
