Better Glucose Control for Patients with Chronic Kidney Disease
ANJ900 has the potential to safely restore metformin use in millions of CKD patients.
Bridging the Treatment Gap
ANJ900 controls blood sugar through a novel gut-targeted mechanism.
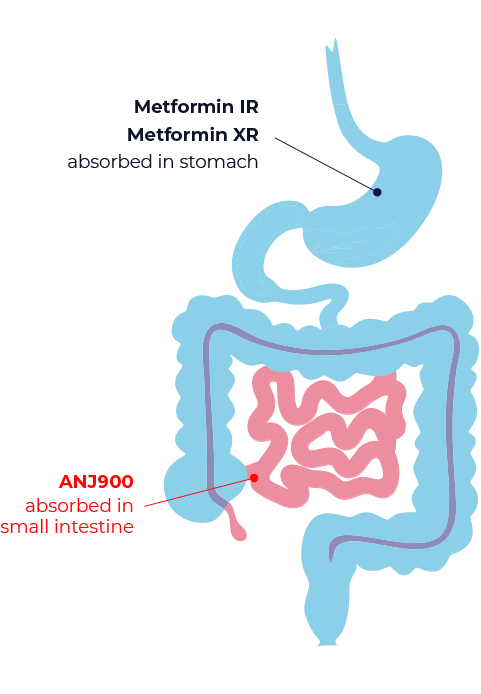
Gut-targeted Therapy Averts Serious Risks
ANJ900 leverages recent insight that the primary site-of-action of metformin, a mainstay of oral type 2 diabetes therapy, is the region of cells lining the gut wall.
With this discovery, the dissolution of ANJ900 has been optimized to deliver metformin only where it’s needed. Gut-targeted therapy avoids unnecessary metformin absorption – a major safety risk for millions of type 2 diabetes patients with chronic kidney disease.
ANJ900 Trials
In previous clinical trials, ANJ900 was shown to be safe and effective for glycemic control in type 2 diabetes patients. Measured levels of metformin in blood were roughly one-third the concentration achieved using conventional formulations and still showed clinically-meaningful glucose lowering effects. These results encouraged us to further investigate the potential of ANJ900 in T2D patients with chronic kidney disease, where full use of metformin is contraindicated.
DREAM-T2D is an ongoing multi-national Phase 3 study designed to show HbA1c lowering in T2D patients with moderate CKD (1-3B) and will utilize a 505(b)(2) pathway for FDA approval. Patients in this trial will receive ANJ900 once daily as oral medication for 28 weeks, and then be eligible for an additional 24 weeks of open label extension where the safety, efficacy, and tolerability of ANJ900 can be further monitored.
